Something missing
Case provided by Thomas Cook 3rd rock ultrasound (emergencyultrasound.com)
Have you ever experienced a time when you were putting a piece of furniture or a puzzle together, and found that a part was missing? Quite frustrating. Well, this may also occur when God puts our heart together. Read on and find out which part was missing in this 14-year-old boy.
A young boy in the emergency room
A 14-year-old boy with palpitations was admitted to a US hospital. He was monitored, but no arrhythmia was found. Take a look at his ECG: Is it a normal ECG for a young boy?
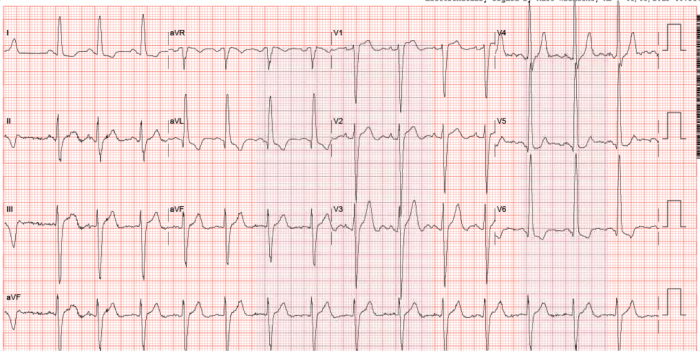 The ECG of a young boy: left axis deviation, left ventricular hypertrophy and abnormal repolarization.
The ECG of a young boy: left axis deviation, left ventricular hypertrophy and abnormal repolarization.
The ECG is quite abnormal. It is typical for the pathology we will show you. Read on.
Echo - what else?
Anyone would perform an echo at this stage, so this was exactly what we did:
 Parasternal long-axis view. Not entirely normal.
Parasternal long-axis view. Not entirely normal.
Do you see anything abnormal or missing here? I would say the ventricle looks enlarged and quite hyperdynamic. However, we need to obtain additional views in order to establish the diagnosis.
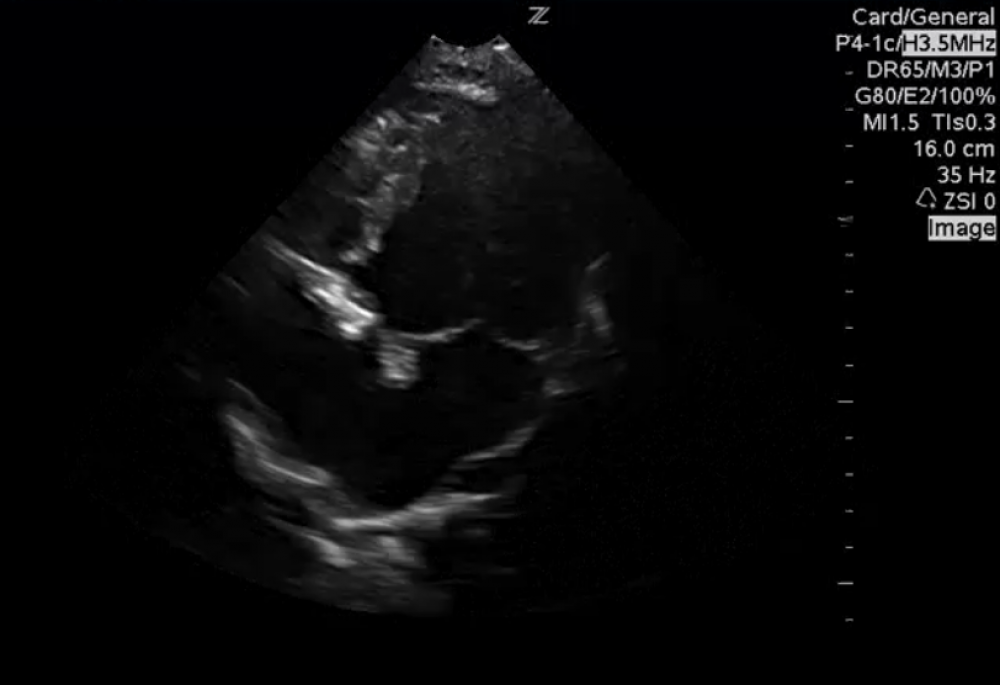 Enlarged left ventricle (and atrium), small right ventricle.
Enlarged left ventricle (and atrium), small right ventricle.
The left ventricle is truly enlarged. In these situations one must investigate the cause of the condition. Possible causes could be mitral or aortic regurgitation, but none of these defects were as pronounced as to cause ventricular dilatation. Can you find a reason?
The missing link
Maybe something is missing. Here is a closer look to help you:
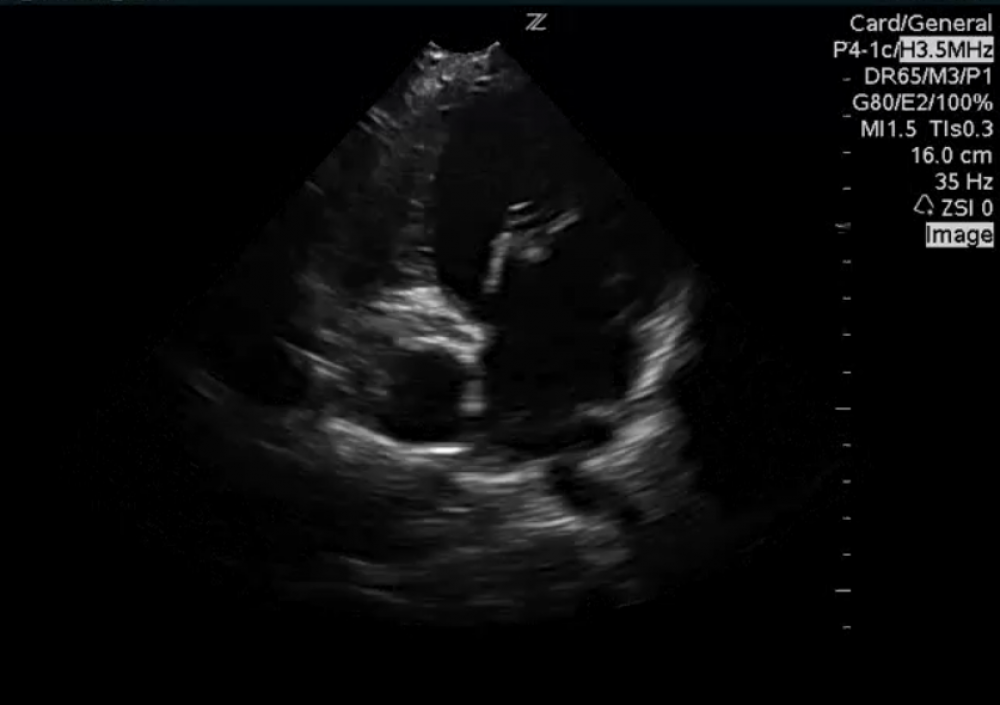 A missing valve.
A missing valve.
The patient has no tricuspid valve. He has a solid sheet of tissue instead. The diagnosis is now clear: tricuspid atresia. This explains why the right atrium is so small (hypoplastic).
Why is he still alive?
We have to understand how the missing tricuspid valve affects circulation. How does blood from the right atrium reach the pulmonary circulation in the absence of a tricuspid valve? The next loop will show you.
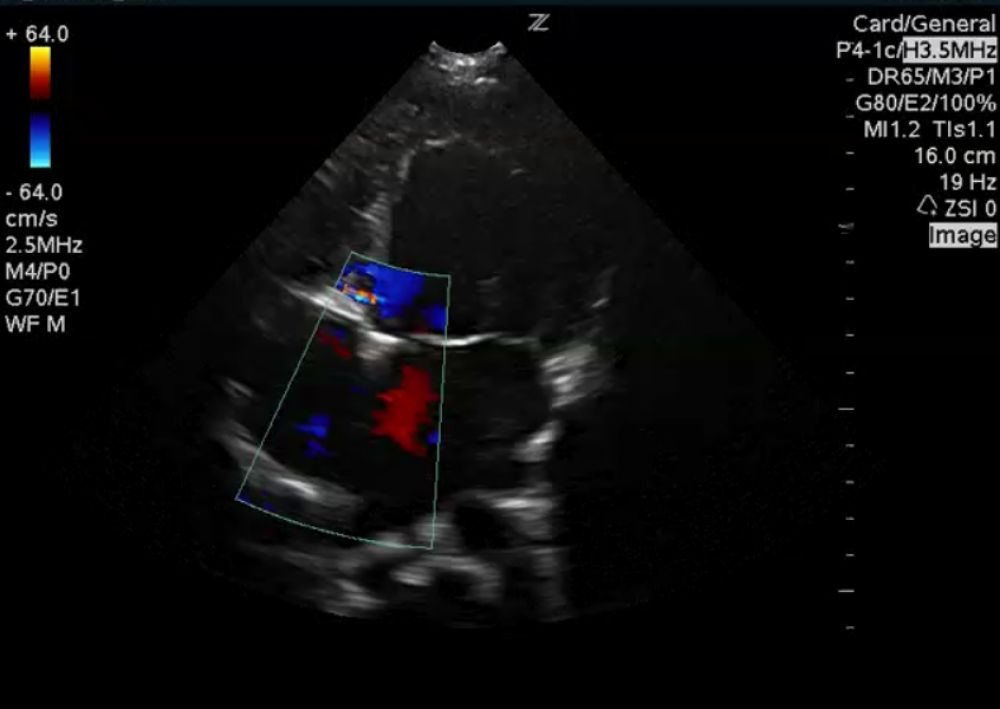 Large atrial septal defect.
Large atrial septal defect.
There is free flow between the atria in the form of a large ASD. Patients with tricuspid atresia must have some form of a right-to-left shunt at the atrial level. Desaturated blood from the right atrium enters the left ventricle. However, this still does not explain how blood reaches the pulmonary artery. It needs something more.
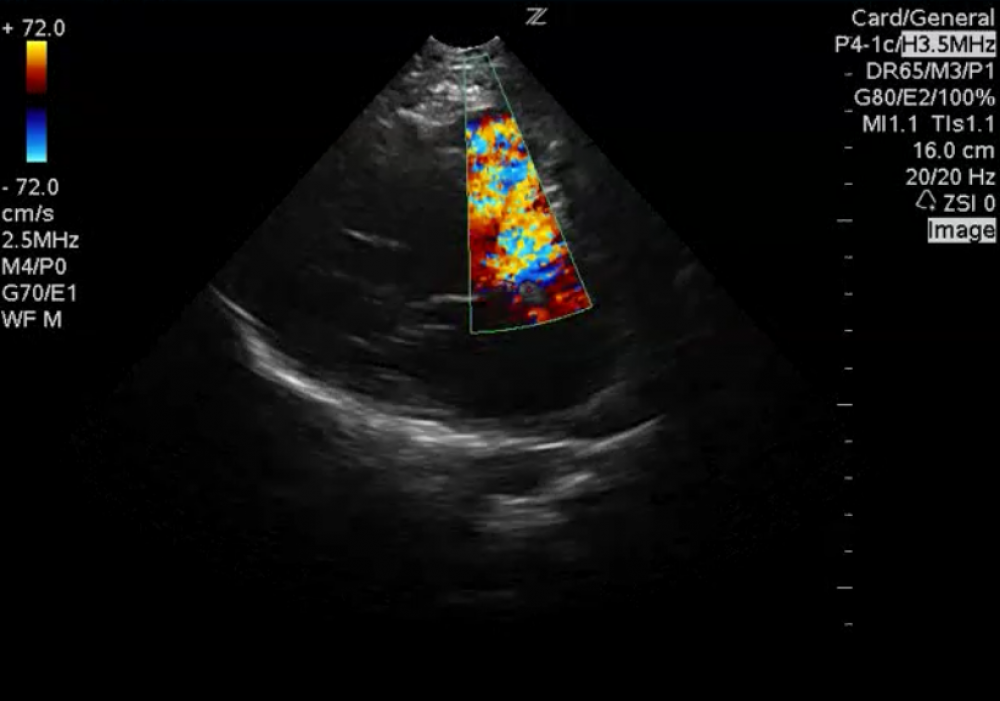 Ventricular septal defect.
Ventricular septal defect.
The parasternal long-axis view shows that a VSD is also present. This allows some blood to reach the pulmonary circulation. When no VSD is present, the patient must at least have a patent ductus arteriosus so that some blood reaches the pulmonary arteries.
Blue baby
As unsaturated blood mixes with saturated blood in the left ventricle, the patient must also be cyanotic. Many patients with tricuspid atresia have some type of pulmonary outflow tract obstruction. Thus, no large quantity of blood flows into the lung (pulmonary overcirculation). This is actually beneficial, because it prevents excessive pulmonary hypertension and the Eisenmenger syndrome. It also dictates the degree of cyanosis. This obstruction may be caused by a rather small VSD (as in this case), or because the patient has a small right ventricular outflow tract, pulmonary stenosis, or hypoplasia of the pulmonary artery. Based on these findings, our patient's condition could be classified as type Ib tricuspid atresia.
Here is the classification scheme if you want to delve deeper into the matter:

Can we fix the problem?
Well, not completely. We have no means of replacing the missing piece in the puzzle (implant a tricuspid valve into the heart). Surgical correction is palliative in nature and is usually performed in several stages. What needs to be done also depends on the defects the patient has and how much blood reaches the pulmonary circulation. Our patient was extremely lucky to reach the age of 14 years. Pulmonary obstruction must have been well balanced: not too much and not too little blood. Most patients require some modification at birth, such as pulmonary banding or a Blalock-Taussig shunt (connection between the innominate/ascending aorta and the central pulmonary artery). Later in life, an attempt is then made to separate the pulmonary circulation from the systemic circulation. This is achieved first by using a Glenn shunt (anastomosis between the superior vena cava and the pulmonary artery), and then performing a Fontan procedure by which blood from the inferior vena cava is also directed towards the pulmonary artery.
Next step
Back to our patient: his palpitation was attributed to anxiety and hyperdynamic heart function. He was moderately cyanotic, but his exercise capacity was rather poor. So the next step was to check his pulmonary pressures by performing a catheter study and determining the type of surgery he needed.
I hope this case has provided some “missing” knowledge. If you liked it, don’t hesitate to forward the link to your friends.
Best,
Thomas
PS: If you missed some of our blog posts just visit our blog!
PPS: If you know someone who might be interested in this case, please forward it to them.



